"I have not yet lost a feeling of wonder, and of delight, that this delicate motion should reside in all the things around us, revealing itself only to him who looks for it. I remember, in the winter of our first experiments, just seven years ago, looking on snow with new eyes. There the snow lay around my doorstep — great heaps of protons quietly precessing in the earth's magnetic field. To see the world for a moment as something rich and strange is the private reward of many a discovery."
-Edward M. Purcell (1952 Nobel Prize in Physics) on the discovery of NMR.
Dynamic nuclear polarization
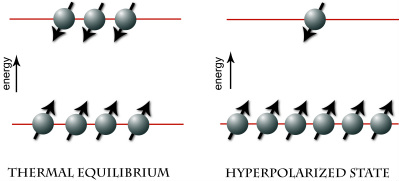
Spin distribution at thermal equilibrium and hyperpolarized states.
Dynamic nuclear polarization (DNP) is a technique that transfers the high electron polarization (owing to their enormous gyromagnetic ratio γ~28,000 MHz/T) to the nuclear spins at low temperature (~1 K) and high magnetic field (>1 T) via microwave irradiation of the sample near the ESR frequency of the paramagnetic electron. The DNP technique, known since the 1950s, has been used in the production of polarized target experiments for nuclear and particle physics experiments. This technology gained renewed impetus with the invention of the dissolution method when the polarized frozen sample is rapidly dissolved with solvents such as water and owing to the long spin-lattice relaxation time T1 of the nucleus of interest, most of the polarization is preserved in the liquid state. These hyperpolarized states typically correspond to >10,000-fold enhancement of the NMR signal at room temperature, allowing high resolution nuclear magnetic resonance spectroscopy (MRS) and imaging (MRI) of insensitive nuclei such as 13C, 15N, 6Li, 89Y, 107,109Ag, etc.--nuclei which were hitherto very difficult or nearly impossible to detect by NMR with 1 scan in the millimolar concentration range.
DNP can be traced in 1953 when Albert Overhauser postulated that the saturation of the ESR line of the conduction electrons leads to an enhancement of the NMR signal. Subsequent experiments by Carver and Slichter in 1956 confirmed the existence of Overhauser effect in metals. The Overhauser effect was extended to non-metals in the works of Abragam et. al. and subsequently the solid effect and thermal mixing DNP mechanisms were discovered. Since the mechanism of DNP is mostly expressed in thermodynamic language, it is important to introduce the concepts of (i) lattice temperature TL and (ii) spin temperature Ts. TL is the thermodynamic temperature of the sample as measured by a sensor, while Ts is the temperature of the spin system calculated using the NMR intensity as the thermometer. Thus Ts is an "artificial" temperature that describes distribution of spin populations in Zeeman energy levels. The time it takes for the spin temperature of the spin system to equalize with the lattice temperature, that is, for the spin system to return to thermal equilibrium, is given by the spin-lattice relaxation time T1. Typically for dissolution DNP, the liquid-state T1 of hyperpolarized substrates need to be at least 10 s so that MRS and MRI experiments could be performed.
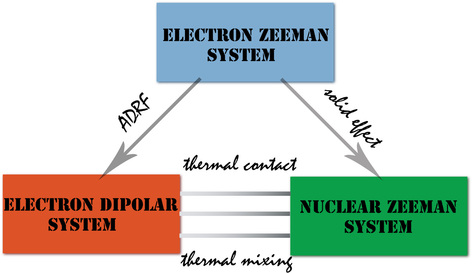
In DNP, the spin temperature of the nuclear spin system is lowered by microwave irradiation. It is important to realize the interplay of three heat reservoirs in DNP: (i) electron Zeeman system (ii) electron spin-spin interaction reservoir and (iii) nuclear Zeeman system. The nuclear spin-spin/dipole-dipole system here was neglected because of its weaker contribution.
DNP can be traced in 1953 when Albert Overhauser postulated that the saturation of the ESR line of the conduction electrons leads to an enhancement of the NMR signal. Subsequent experiments by Carver and Slichter in 1956 confirmed the existence of Overhauser effect in metals. The Overhauser effect was extended to non-metals in the works of Abragam et. al. and subsequently the solid effect and thermal mixing DNP mechanisms were discovered. Since the mechanism of DNP is mostly expressed in thermodynamic language, it is important to introduce the concepts of (i) lattice temperature TL and (ii) spin temperature Ts. TL is the thermodynamic temperature of the sample as measured by a sensor, while Ts is the temperature of the spin system calculated using the NMR intensity as the thermometer. Thus Ts is an "artificial" temperature that describes distribution of spin populations in Zeeman energy levels. The time it takes for the spin temperature of the spin system to equalize with the lattice temperature, that is, for the spin system to return to thermal equilibrium, is given by the spin-lattice relaxation time T1. Typically for dissolution DNP, the liquid-state T1 of hyperpolarized substrates need to be at least 10 s so that MRS and MRI experiments could be performed.
In DNP, the spin temperature of the nuclear spin system is lowered by microwave irradiation. It is important to realize the interplay of three heat reservoirs in DNP: (i) electron Zeeman system (ii) electron spin-spin interaction reservoir and (iii) nuclear Zeeman system. The nuclear spin-spin/dipole-dipole system here was neglected because of its weaker contribution.
Calculating the polarization
|
DNP Instrumentation
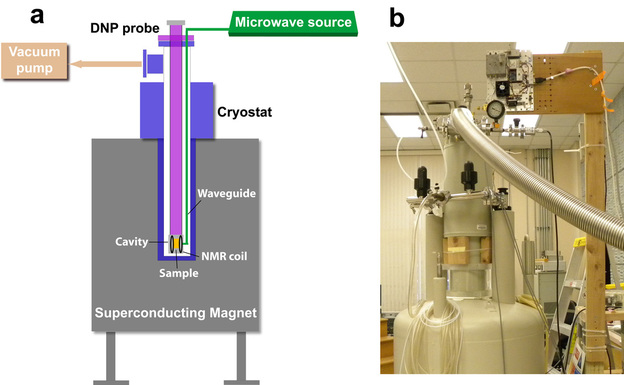 (a) Schematic diagram of the DNP polarizer. (b) Photo of the 129 GHz homebuilt DNP polarizer at UT Southwestern Medical Center.
(a) Schematic diagram of the DNP polarizer. (b) Photo of the 129 GHz homebuilt DNP polarizer at UT Southwestern Medical Center.
The instrumentation for dynamic nuclear polarization (DNP), a technique that amplifies the nuclear polarization, has been known since the 1960s. Shown on the right is a modified version designed for dissolution DNP for biomedical applications. Generally, the main parts of a DNP polarizer are: (1) superconducting magnet (2) cryostat (3) microwave source (4) DNP insert (5) vacuum system (6) NMR detection circuit (7) dissolution system.
How to achieve the maximum DNP-NMR signal via thermal mixing
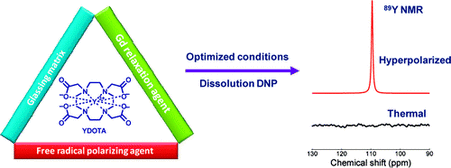
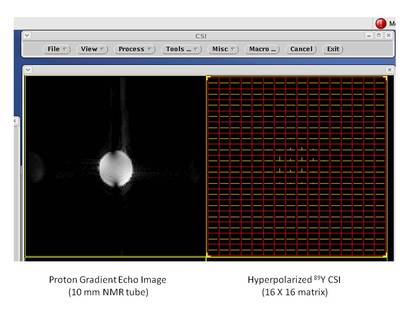
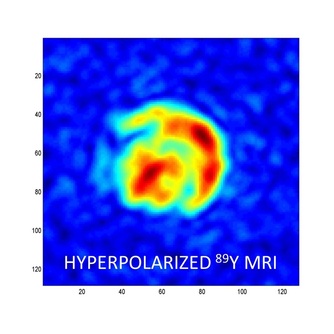
From Lumata et al., J. Am. Chem. Soc. 133, 8673-8680 (2011): "Hyperpolarized 89Y complexes are attractive NMR spectroscopy and MR imaging probes due to the exceptionally long spin–lattice relaxation time (T1 ≈ 10 min) of the 89Y nucleus. However, in vivo imaging of 89Y has not yet been realized because of the low NMR signal enhancement levels previously achieved for this ultra low-γn nucleus. Here, we report liquid-state 89Y NMR signal enhancements over 60 000 times the thermal signal at 298 K in a 9.4 T magnet, achieved after the dynamic nuclear polarization (DNP) of Y(III) complex of 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) samples at 3.35 T and 1.4 K. The 89Y DNP was shown to proceed by thermal mixing and the liquid state 89Y NMR signal enhancement was maximized by (i) establishing the optimal microwave irradiation frequency, (ii) optimizing the glassing matrix, (iii) choosing a radical with negligible inhomogeneous line broadening contribution to the ESR linewidth, and (iv) addition of an electron T1e relaxation agent. The highest enhancements were achieved using a trityl OX063 radical combined with a gadolinium relaxation agent in water-glycerol matrix. Co-polarization of 89YDOTA and sodium [1-13C]pyruvate showed that both 89Y and 13C nuclear species acquired the same spin temperature, consistent with thermal mixing theory of DNP. This methodology may be applicable for the optimization of DNP of other low-γn nuclei."
Impact of Gd3+ on 13C dynamic nuclear polarization
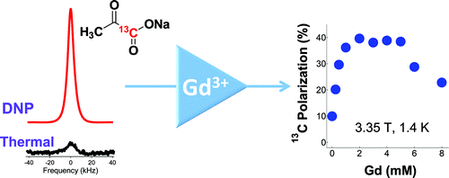
From Lumata et al., J. Phys. Chem A. 116, 5129-5138 (2012): "Hyperpolarized [1-13C]pyruvate has become an important diagnostic tracer of normal and aberrant cellular metabolism for in vitro and in vivo NMR spectroscopy (MRS) and imaging (MRI). In pursuit of achieving high NMR signal enhancements in dynamic nuclear polarization (DNP) experiments, we have performed an extensive investigation of the influence of Gd3+ doping, a parameter previously reported to improve hyperpolarized NMR signals, on the DNP of this compound. [1-13C]Pyruvate samples were doped with varying amounts of Gd3+ and fixed optimal concentrations of free radical polarizing agents commonly used in fast dissolution DNP: trityl OX063 (15 mM), 4-oxo-TEMPO (40 mM), and BDPA (40 mM). In general, we have observed three regions of interest, namely, (i) a monotonic increase in DNP-enhanced nuclear polarization Pdnp upon increasing the Gd3+ concentration until a certain threshold concentration c1 (1–2 mM) is reached, (ii) a region of roughly constant maximum Pdnp from c1 until a concentration threshold c2 (4–5 mM), and (iii) a monotonic decrease in Pdnp at Gd3+ concentration c > c2. Of the three free radical polarizing agents used, trityl OX063 gave the best response to Gd3+ doping, with a 300% increase in the solid-state nuclear polarization, whereas addition of the optimum Gd3+ concentration on BDPA and 4-oxo-TEMPO-doped samples only yielded a relatively modest 5–20% increase in the base DNP-enhanced polarization. The increase in Pdnp due to Gd3+ doping is ascribed to the decrease in the electronic spin–lattice relaxation T1e of the free radical electrons, which plays a role in achieving lower spin temperature Ts of the nuclear Zeeman system. These results are discussed qualitatively in terms of the spin temperature model of DNP."
Influence of deuteration in the glassing matrix on 13C dynamic nuclear polarization
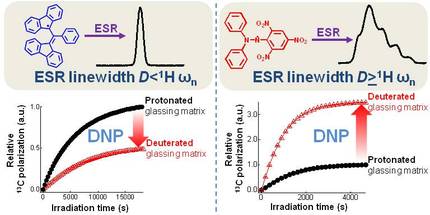
From Lumata et al., Phys. Chem. Chem. Phys 15, 7032-7035 (2013): "Replacement of protons by deuterons in the glassing solvents led to 2-3-fold improvement of the 13C dynamic nuclear polarization (DNP) solid-state NMR signal for samples doped with large electron spin resonance (ESR) linewidth free radicals galvinoxyl, DPPH, and 4-oxo-TEMPO. Meanwhile, the reverse effect is observed for 13C DNP using small ESR linewidth free radicals BDPA and trityl OX063."
It was reported in a previous study that deuteration of the glassing solvents of 13C samples doped with TEMPO free radical approximately doubled the nuclear polarization. Based on this paper, it is becoming customary to use deuterated glassing matrices regardless of the type of free radical polarizing agent. Here we investigate the influence of deuteration of the glassing matrix on 13C DNP samples doped with different free radical polarizing agents commonly used for dissolution DNP: BDPA, trityl OX063, galvinoxyl, DPPH, and 4-oxo-TEMPO.
Results: Deuteration of the glassing matrix increases the 13C polarization of samples doped with free radicals that have ESR D comparable to or larger than the 1H Larmor frequency such as galvinoxyl, DPPH, and 4-oxo-TEMPO. This is attributed to the lower heat capacity of the deuteron Zeeman system compared to protons. However, glassing solvent deuteration is not recommended for DNP with free radicals that have narrow ESR linewidth such as BDPA and trityl OX063 where the proton Zeeman system has little or no contact with EDS. In this case, deuteration would only add more heat load to the nuclear Zeeman system, resulting in lower 13C polarization.
It was reported in a previous study that deuteration of the glassing solvents of 13C samples doped with TEMPO free radical approximately doubled the nuclear polarization. Based on this paper, it is becoming customary to use deuterated glassing matrices regardless of the type of free radical polarizing agent. Here we investigate the influence of deuteration of the glassing matrix on 13C DNP samples doped with different free radical polarizing agents commonly used for dissolution DNP: BDPA, trityl OX063, galvinoxyl, DPPH, and 4-oxo-TEMPO.
Results: Deuteration of the glassing matrix increases the 13C polarization of samples doped with free radicals that have ESR D comparable to or larger than the 1H Larmor frequency such as galvinoxyl, DPPH, and 4-oxo-TEMPO. This is attributed to the lower heat capacity of the deuteron Zeeman system compared to protons. However, glassing solvent deuteration is not recommended for DNP with free radicals that have narrow ESR linewidth such as BDPA and trityl OX063 where the proton Zeeman system has little or no contact with EDS. In this case, deuteration would only add more heat load to the nuclear Zeeman system, resulting in lower 13C polarization.
ESR studies of trityl OX063 at multiple magnetic fields
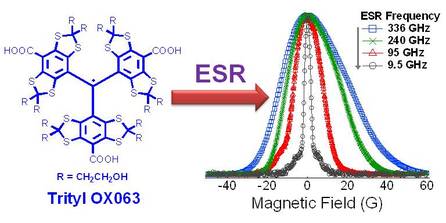
From Lumata et al., Phys. Chem. Chem. Phys. 2013: "We have performed temperature-dependent electron spin resonance (ESR) measurements of the stable free radical trityl OX063, an efficient polarizing agent for dissolution dynamic nuclear polarization (DNP), at the optimum DNP concentration (15 mM). We have found that (i) when compared to the W-band electron spin–lattice relaxation rate of other free radicals used in DNP at the same concentration, trityl OX063 has slower T1e^-1 than BDPA and 4-oxo-TEMPO. At T > 20 K, the T1e^-1 vs. T data of trityl OX063 appears to follow a power law dependence close to the Raman process prediction whereas at T o 10 K, electronic relaxation slows and approaches the direct process behaviour. (ii) Gd3+ doping, a factor known to enhance DNP, of trityl OX063 samples measured at W-band resulted in monotonic increases of T1e^-1 especially at temperatures below 20–40 K while the ESR lineshapes remained essentially unchanged. (iii) The high frequency ESR spectrum can be fitted with an axial g-tensor with a slight g-anisotropy: gx = gy = 2.00319(3) and gz = 2.00258(3). Although the ESR linewidth D monotonically increases with field, the temperature-dependent T1e 1 is almost unchanged as the ESR frequency is increased from 9.5 GHz to 95 GHz, but becomes faster at 240 GHz and 336 GHz. The ESR properties of trityl OX063 reported here may provide insights into the efficiency of DNP of low-gamma nuclei performed at various magnetic fields, from 0.35 T to 12 T."
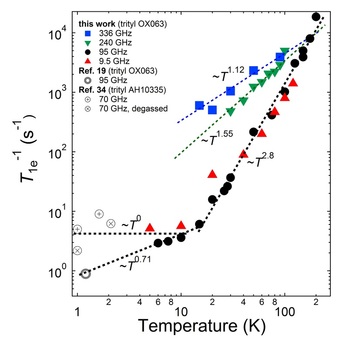
Temperature-dependent electron relaxation rate T1e^-1 of trityl OX063 at different ESR frequencies/magnetic fields from this work and a data point at 1.2 K from ref. 19 (J. H. Ardenkjaer-Larsen, S. Macholl and H. Johannesson, Appl. Magn. Reson., 2008, 34, 509–522). Low temperature NEDOR T1e^-1 data points of a trityl derivative AH10335 (dissolved in butanol) at 70 GHz from ref. 34 (C. Hess, J. Herick, A. Berlin, W. Meyer and G. Reichers, Nucl. Instrum. Methods Phys. Res., Sect. A, 2012, 694, 69–77.) were also plotted for comparison. The dashed lines are fits/extrapolations using power law equations where alpha is the extracted power law exponent.
It is interesting to note that although there is a significant ESR linewidth broadening from
0.35 T (X-band) to 3.35 T (W-band), the temperature dependence of their electron relaxation rates are very similar. The similarity of the relaxation rate values of trityl OX063 (15 mM) at X- and W-bands was also previously observed in an earlier work on a more dilute trityl radical concentration (0.2 mM). This relaxation behavior was ascribed to the field-independent Raman process and local mode being the dominant relaxation processes at a wide temperature range for trityl radical. On the other hand, increasing the magnetic field to 8.5 T (240 GHz) and 12 T (336 GHz) shortens the electron relaxation time. This is in line with the expected frequency dependence of the direct process. Nevertheless, the log–log plot of electron relaxation rate vs. temperature shown in the Figure shows a monotonic decrease in slope in the high temperature region (T > 15 K) as the field is increased from X or W-band, suggesting deviation from the Raman process. As DNP is pushed to higher fields, the ESR properties of the free radical polarizing agent (e.g. linewidth D and spin–lattice T1e relaxation) play an important role in the performance of DNP. The current results seem to suggest that higher field and lower temperatures will continue to provide increases in nuclear polarization which will approach the theoretical limit without serious hindrance from the free radical relaxation properties.
It is interesting to note that although there is a significant ESR linewidth broadening from
0.35 T (X-band) to 3.35 T (W-band), the temperature dependence of their electron relaxation rates are very similar. The similarity of the relaxation rate values of trityl OX063 (15 mM) at X- and W-bands was also previously observed in an earlier work on a more dilute trityl radical concentration (0.2 mM). This relaxation behavior was ascribed to the field-independent Raman process and local mode being the dominant relaxation processes at a wide temperature range for trityl radical. On the other hand, increasing the magnetic field to 8.5 T (240 GHz) and 12 T (336 GHz) shortens the electron relaxation time. This is in line with the expected frequency dependence of the direct process. Nevertheless, the log–log plot of electron relaxation rate vs. temperature shown in the Figure shows a monotonic decrease in slope in the high temperature region (T > 15 K) as the field is increased from X or W-band, suggesting deviation from the Raman process. As DNP is pushed to higher fields, the ESR properties of the free radical polarizing agent (e.g. linewidth D and spin–lattice T1e relaxation) play an important role in the performance of DNP. The current results seem to suggest that higher field and lower temperatures will continue to provide increases in nuclear polarization which will approach the theoretical limit without serious hindrance from the free radical relaxation properties.
Galvinoxyl as a polarizing agent for dissolution DNP-NMR spectroscopy
From Lumata et al. J. Magn. Reson. 227, 14-19 (2013): "The goal of this work was to test feasibility of using galvinoxyl (2,6-di-tert-butyl-α-(3,5-di-tert-butyl-4-oxo-2,5-cyclohexadien-1-ylidene)-p-tolyloxy) as a polarizing agent for dissolution dynamic nuclear polarization (DNP) NMR spectroscopy. We have found that galvinoxyl is reasonably soluble in ethyl acetate, chloroform, or acetone and the solutions formed good glasses when mixed together or with other solvents such as dimethyl sulfoxide. W-band electron spin resonance (ESR) measurements revealed that galvinoxyl has an ESR linewidth D intermediate between that of carbon-centered free radical trityl OX063 and the nitroxide-based 4-oxo-TEMPO, thus the DNP with galvinoxyl for nuclei with low gyromagnetic ratio γ such as 13C and 15N is expected to proceed predominantly via the thermal mixing process. The optimum radical concentration that would afford the highest 13C nuclear polarization (approximately 6% for [1-13C]ethyl acetate) at 3.35 T and 1.4 K was found to be around 40 mM. After dissolution, large liquid-state NMR enhancements were achieved for a number of 13C and 15N compounds with long spin–lattice relaxation time T1. In addition, the hydrophobic galvinoxyl free radical can be easily filtered out from the dissolution liquid when water is used as the solvent. These results indicate that galvinoxyl can be considered as an easily available free radical polarizing agent for routine dissolution DNP-NMR spectroscopy."
The efficiency of DPPH as a polarizing agent for DNP-NMR spectroscopy
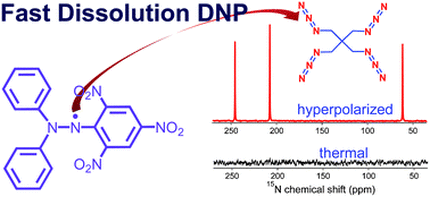
From Lumata et al., RSC Adv. 2, 12812-12817 (2012): "The free radical 2,2-diphenyl-1-picrylhydrazyl (DPPH) was tested as a polarising agent for fast dissolution dynamic nuclear polarisation (DNP) NMR spectroscopy. DPPH was found to be reasonably soluble in sulfolane and the optimum concentration for DNP is 20–40 mM depending upon whether short polarisation times or the maximum signal intensity is needed. W-band ESR measurements revealed that the ESR linewidth D of DPPH is intermediate between that of BDPA and 4-oxo-TEMPO. Several thousand-fold NMR signal enhancements in the liquid-state were achieved for 13C, 15N, 89Y, and 109Ag compounds, demonstrating that DPPH can be added to the list of polarising agents for DNP-NMR spectroscopy. Furthermore, the hydrophobic DPPH free radical can be easily filtered out from the dissolution liquid when water is used as the dissolution solvent."
BDPA: teaching an old free radical new tricks
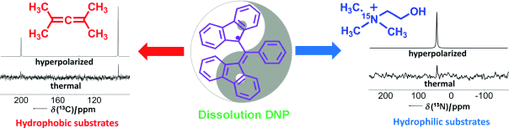
From Lumata et al., Chem. Eur. J. 17, 10825-10827 (2011): "The stable free radical 1,3-bisdiphenylene-2-phenylallyl (BDPA) is an efficient polarizing agent in the dissolution dynamic nuclear polarization (DNP) of a variety of hydrophilic and hydrophobic compounds, such as tetramethylallene or [15N]choline (see figure). In addition, BDPA can be easily removed from the hyperpolarized solution by a simple mechanical filtration, when water is used for dissolution."
The two lives of hyperpolarized Silver
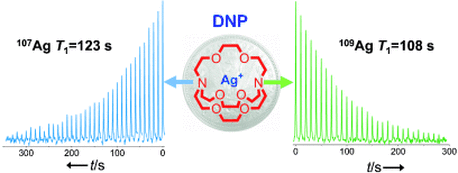
From Lumata et al., Angew. Chem. Intl. Ed. 51, 525-527 (2012): Both isotopes of silver, 107Ag and 109Ag, were simultaneously polarized by dynamic nuclear polarization (DNP), thus allowing large signal enhancements and the NMR characterization of Ag complexes in the millimolar concentration range. Since both isotopes have long relaxation times T1, the hyperpolarized NMR signal of one isotope could still be observed even after the magnetization of the other isotope had been destroyed by radio-frequency pulses.
Physics of Hyperpolarized [13C]-89YDOTA
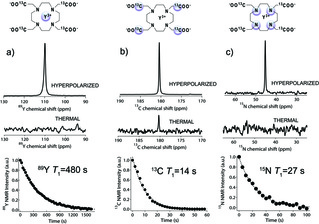
From Lumata et al., Appl. Magn. Reson. 43, 69-79 (2012): "The yttrium complex of 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetra(1′-13C-acetic acid) [13C]DOTA was synthesized. Fast dissolution dynamic nuclear polarization (DNP) nuclear magnetic resonance (NMR) studies revealed that the 89Y, 13C, and 15N nuclei present in the complex could be co-polarized at the same optimum microwave irradiation frequency. The liquid-state spin–lattice relaxation time T 1 of these nuclei were found to be reasonably long to preserve some or most of the DNP-enhanced polarization after dissolution. The hyperpolarized 13C and 89Y NMR signals were optimized in different glassing mixtures. The overall results are discussed in light of the thermal mixing model of DNP."
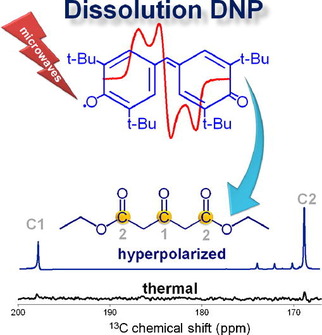
13C spin diffusion: domino effect expedites DNP process
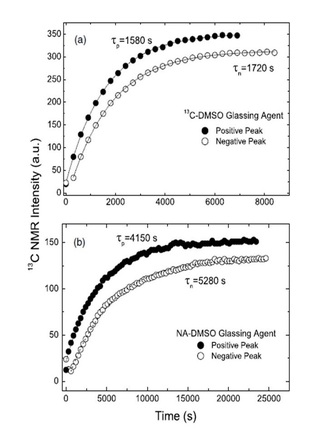
From Lumata et al., Phys. Med. Biol. 56, N85-N92 (2011): "Dimethyl sulfoxide (DMSO) can effectively form a glassy matrix necessary for dynamic nuclear polarization (DNP) experiments. We tested the effects of 13C enrichment in DMSO on DNP of [1-13C]pyruvate doped with trityl radical OX063Me. We found that the polarization build-up time τ of pyruvate in 13C-labeled DMSO glassing solution is twice as fast as the unenriched DMSO while the nuclear magnetic resonance enhancement was unchanged. This indicates that 13C–13C spin diffusion is a limiting factor in the kinetics of DNP in this system, but it has a minimal effect on the absolute value of polarization achievable for the target."
"Dynamic nuclear polarization has recently been used to produce highly polarized solutions of 13C nuclei suitable for MRI studies. In this report, we detail the use of 13C DMSO in the glassing matrix used to polarize [1-13C]pyruvate. Inclusion of the labeled DMSO results in a faster build up rate for the pyruvate, which can be attributed to increased 13C–13C spin diffusion in the sample."
"Dynamic nuclear polarization has recently been used to produce highly polarized solutions of 13C nuclei suitable for MRI studies. In this report, we detail the use of 13C DMSO in the glassing matrix used to polarize [1-13C]pyruvate. Inclusion of the labeled DMSO results in a faster build up rate for the pyruvate, which can be attributed to increased 13C–13C spin diffusion in the sample."
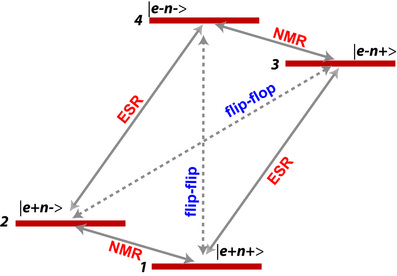
Thermal mixing
Thermal mixing occurs when the electronic linewidth δωe is greater than the nuclear Larmor frequency νn. In this case, the nuclear Zeeman system (nZS) is in thermal contact with the electron spin-spin interaction system (eSSIS). In other words, both nZS and eSSIS are "on resonance" and have comparable energies thus they can exchange energies. This leads to the equalization of the spin temperature of both systems under microwave irradiation.
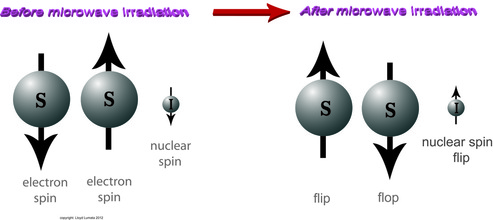
The Solid Effect
DNP: application of physics to chemistry and biology
DNP, a technology emanating from nuclear and particle physics [1-3], has found renaissance in chemical and biomedical NMR research with the invention of the dissolution method [4] which brings the NMR signal amplification capability of DNP to liquid-state NMR at physiological temperatures. Hyperpolarized 13C NMR/MRI [5-7] are utilized to monitor metabolic activities of normal and aberrant cells/tissues in real-time with the potential of providing important diagnostic information for a much-needed targeted therapeutic intervention. In chemistry, dissolution DNP has found routine use in structural characterization of compounds by NMR with superb sensitivity [8] and monitoring chemical reactions with sub-second time resolution. As such, DNP, which is a combination of ESR, NMR, and cryogenic technologies, is an example of science that transcends barriers, extending its applications from physics to chemistry, materials science, and biomedical research. Here are some of target applications of dissolution DNP:
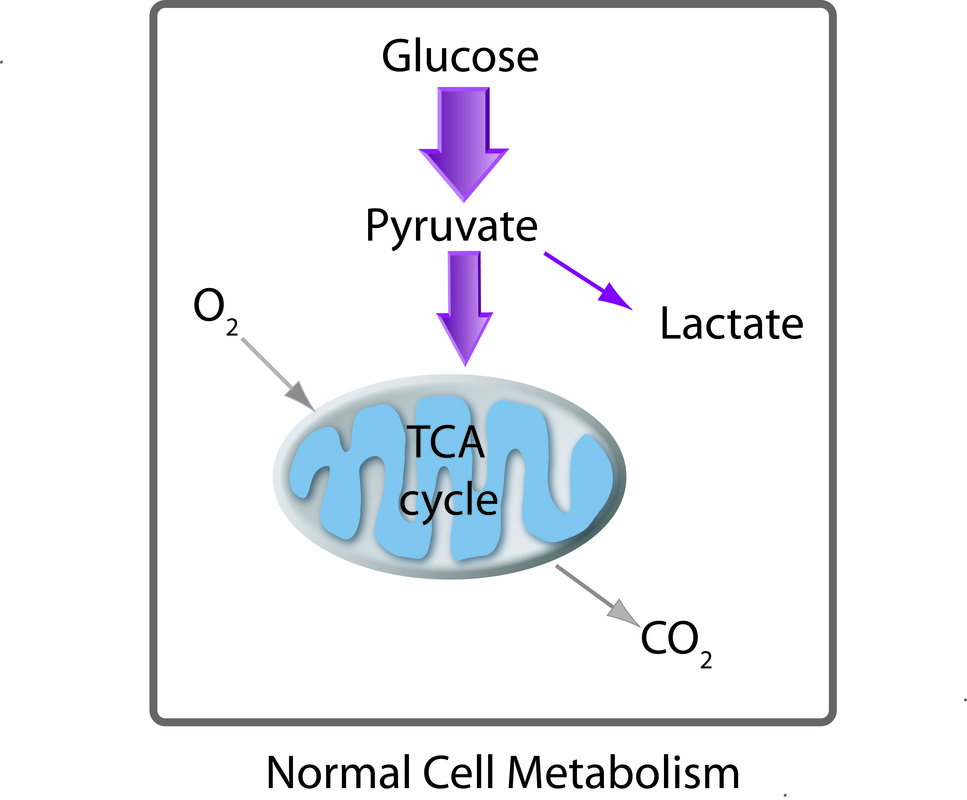
Glycolysis
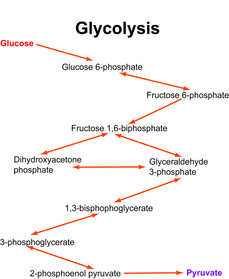 Production of pyruvate from glucose.
Glycolysis is a series of 10 enzyme-catalyzed reactions that converts glucose into pyruvate. Glycolysis occurs in the cytosol of the cell. Note that enzymes involved in the reaction steps are currently not shown in this figure. Click here for a detailed schematic of glycolytic pathways. An excellent animation of glycolysis is provided by the International Union of Biochemistry and Molecular Biology (IUBMB, click here for the link to the animation). Aberrant glycolytic activity of cells in diseases such as diabetes and cancer are important to understand and carbon-13 NMR is an excellent tool to elucidate the abnormal glycolytic activities. The problem with carbon-13 NMR is the insensitivity but this has recently been addressed by dissolution DNP-NMR technology with allows real time monitoring of metabolic activities via 13C NMR with superb sensitivity.
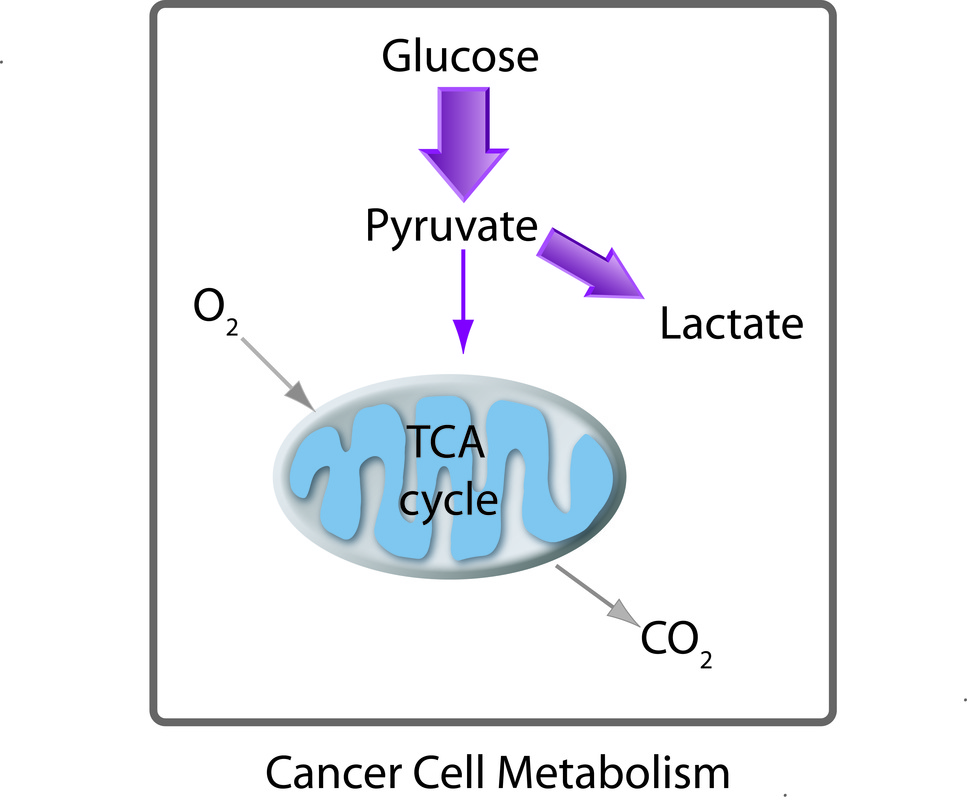
Normal and Aberrant Cell Metabolism
|
TCA Cycle
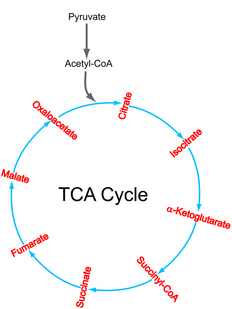 A simplified version of the TCA cycle.
The tricarboxylic acid (TCA) cycle, also known as citric acid or Krebs cycle, is a series of enzyme-catalyzed reactions that oxidizes acetyl-CoA from pyruvate (the end product of glycolysis), reduces NAD+ into NADH and produces carbon dioxide. NADH is prerequisite to the production of ATP, the energy currency of cells. The TCA cycle occurs in the mitochondria for eukaryotic cells and in the cytosol for bacteria. Note: the backward reaction of the most of the steps in the TCA cycle are not shown. Click here for a more detailed TCA cycle diagram. The TCA cycle is a central series of biochemical reactions for all aerobic organisms, thus an important target of the hyperpolarized carbon-13 NMR technology which is being utilized to understand normal and aberrant cellular metabolism.
|
References:
[1] A. Abragam and M. Goldman, Rep. Prog. Phys. 41, 395-467 (1978).
[2] W. de Boer, J. Low Temp. Phys. 22, 185-212 (1976).
[3] D. G. Crabb and W. Meyer, Annu. Rev. Nucl. Part. Sci. 47, 67-109 (1997).
[4] J. H. Ardenkjaer-Larsen et al., Proc. Natl. Acad. Sci. U.S.A. 100, 10158-10163 (2003).
[5] J. Kurhanewicz et al., Neoplasia 13, 81-97 (2011).
[6] K. Brindle, Nat. Rev. Cancer 8, 94-107 (2008).
[7] K. M. Brindle et al., Magn. Reson. Med. 66, 505-519 (2011).
[8] L. Lumata et. al., Angew. Chem. Intl. Ed. 51, 525-527 (2012).
[9] M. G. Vander Heiden, L. C. Cantley, and C. B. Thompson, "Understanding the Warburg effect: the metabolic requirements of cell proliferation" Science 324, 1029-1033 (2009).
[1] A. Abragam and M. Goldman, Rep. Prog. Phys. 41, 395-467 (1978).
[2] W. de Boer, J. Low Temp. Phys. 22, 185-212 (1976).
[3] D. G. Crabb and W. Meyer, Annu. Rev. Nucl. Part. Sci. 47, 67-109 (1997).
[4] J. H. Ardenkjaer-Larsen et al., Proc. Natl. Acad. Sci. U.S.A. 100, 10158-10163 (2003).
[5] J. Kurhanewicz et al., Neoplasia 13, 81-97 (2011).
[6] K. Brindle, Nat. Rev. Cancer 8, 94-107 (2008).
[7] K. M. Brindle et al., Magn. Reson. Med. 66, 505-519 (2011).
[8] L. Lumata et. al., Angew. Chem. Intl. Ed. 51, 525-527 (2012).
[9] M. G. Vander Heiden, L. C. Cantley, and C. B. Thompson, "Understanding the Warburg effect: the metabolic requirements of cell proliferation" Science 324, 1029-1033 (2009).